Purpose and function of surface treatment of machined parts:The purpose of surface treatment of CNC machining parts is to achieve corrosion resistance, wear resistance, beauty and improve service life.AN-Prototype has many years of rich experience in a series of services from parts processing to surface treatment to assembly. In addition to CNC technology, it also has very rich experience in surface treatment.The existing surface treatment process covers: painting, baking, powder spraying, sandblasting, shot blasting, anodizing, thick film oxidation, micro-arc oxidation, electroplating, electrophoresis, laser engraving, silk screen printing, wire drawing, mirror polishing, dyeing, blackening , CD pattern, etching, high gloss, etching pattern, glue dropping, etc.,

Table of Contents
ToggleAnodizing
It is an electrolytic oxidation process, which converts the surface of the material into a protective film, making it difficult to oxidize and corrode, prolonging the life and achieving the appearance of various colors. Commonly used oxidation treatments are divided into: ordinary Anodizing, wire drawing oxidation, hard oxidation, thick film oxidation, micro-arc oxidation, etc. The materials that can be oxidized are: aluminum alloy, magnesium alloy, titanium alloy, etc.
Aluminum alloy processing parts will be oxidized after a long time in the air. The oxide film naturally formed on the aluminum surface is amorphous, which will make the aluminum metal surface lose its original luster. After Anodizing treatment, the aluminum parts processed by CNC A layer of dense film much thicker than the natural oxide film is obtained on the surface. After this layer of artificial oxide film is sealed, the amorphous oxide film is transformed into a crystalline oxide film, and the pores are also closed, so that the metal surface gloss can be maintained for a long time. It is necessary to anodize aluminum alloy parts. of.
The characteristics of anodized aluminum parts are as follows:
a. Prevent surface corrosion of CNC processed aluminum products, improve service life and structural stability:
Since the film layer obtained by Anodizing itself has sufficient stability in the atmosphere, the oxide film on the aluminum surface can be used as a protective layer, which can effectively protect the surface of aluminum products from corrosion and prolong the service life.
b. CNC Machining aluminum products for anodizing can play a decorative role:
For most CNC Machined aluminum alloy products that require surface decoration, after chemical or electrochemical polishing, Anodizing with sulfuric acid solution can obtain an oxide film with high transparency. This oxide film can absorb many kinds of organic dyes and inorganic dyes, so it has a variety of bright colors. This layer of colored film is not only an anti-corrosion layer, but also a decorative layer, which is usually called coloring treatment. Under some special process conditions, a protective and decorative oxide film similar in appearance to porcelain can also be obtained. The oxidation treatment colors of aluminum products made by daily processing plants are black, silver, blue, red, golden yellow, etc. The color is selected according to the user’s designation.
c. Anodizing treatment can improve the insulation of CNC Machined aluminum products:
The oxide film obtained after Anodizing of aluminum and aluminum alloy products has a large resistance, so it has a certain effect on improving the electrical insulation of CNC aluminum structural parts. Moreover, the anodized oxidation process also improves the bonding force with the organic coating and improves the bonding force with the inorganic coating layer.
d. Hard oxidation treatment of aluminum products can improve its mechanical properties:
The pores and absorption properties of the film layer are used to store the selected oil, which is effectively applied to the working conditions under the friction state, and has the characteristics of lubrication and wear resistance.

Plating
Electroplating is the process of plating a thin layer of other metals or alloys on the surface of metal parts using the principle of electrolysis. It is a process of using electrolysis to attach a layer of metal film to the surface of metal or other materials to prevent metal oxidation (such as Rust), improve wear resistance, conductivity, reflectivity, corrosion resistance (copper sulfate, etc.) and enhance the appearance, etc. will make your products more high-end fashion and bring a better market.
Plating method
Electroplating is divided into rack plating, barrel plating, continuous plating and brush plating, which are mainly related to the size and batch size of the parts to be plated. Rack plating is suitable for general-sized products, such as car bumpers, bicycle handlebars, etc. Barrel plating is suitable for small parts, fasteners, washers, pins, etc. Continuous plating is suitable for mass-produced wire and strip. Brush plating is suitable for partial plating or restoration. The electroplating solution includes acidic, alkaline, acidic and neutral solutions with chromium admixture. No matter what kind of plating method is used, the plating tanks and hanging tools that are in contact with the products to be plated and the plating solution should have a certain degree of safety. Versatility.
Coating Classification
According to the composition of the coating, it can be divided into three types: single metal coating, alloy coating and composite coating.If classified according to the purpose, it can be divided into:
a. protective coating;
b. protective decorative coating;
c. decorative coating;
d. Restorative coating;
e. functional coating
Single metal plating
Single-metal electroplating has a history of more than 170 years, and 33 metals on the periodic table can be prepared from aqueous solution by electrodeposition. There are more than 10 kinds of electroplating zinc, nickel, chromium, copper, tin, iron, cobalt, cadmium, lead, gold, silver, etc. The coating formed by simultaneously depositing two or more elements on the cathode is an alloy coating. The alloy coating has the structure and properties that a single metal coating does not have, such as amorphous Ni-P alloy, sn alloy of each core that is not on the phase diagram, and has a special decorative appearance, particularly high corrosion resistance and excellent weldability , Magnetic alloy coating, etc.
Composite plating
Composite plating is a process in which solid particles are added to the plating solution to co-deposit with metals or alloys to form a metal-based surface composite material to meet special application requirements. According to the classification of electrochemical properties between the coating and the base metal, the electroplating coating can be divided into two categories: anodic coating and cathodic coating. When the potential of the coating metal relative to the base metal is negative, the coating is an anode when a corrosion micro-battery is formed, so it is called an anodic coating, such as the galvanized layer on a steel piece; and when the potential of the coating metal relative to the base metal is positive, When the corrosion micro-battery is formed, the coating is the cathode, so it is called the cathodic coating, such as the nickel-plated layer and tin-plated layer on steel parts.
Classification by use can be divided into:
①Protective coating: coatings such as Zn, Ni, Cd, Sn and Cd-Sn are used as anti-corrosion coatings that are resistant to atmosphere and various corrosive environments;
② Protection. Decorative coating: such as Cu-Ni-Cr, Ni-Fe-Cr composite coating, etc., which are both decorative and protective;
③Decorative coating: such as Au, Ag and Cu. Sun imitation gold plating, black chrome, black nickel plating, etc.;
④ Restorative coating: such as electroplating Ni, Cr, Fe layer to repair some expensive wearable parts or processing out-of-tolerance parts;
⑤Functional coatings: conductive coatings such as Ag and Au; magnetic coatings such as Ni-Fe, Fe-Co, Ni-Co; high-temperature anti-oxidation coatings such as Cr and Pt-Ru; Anti-reflective coatings such as black nickel; hard chrome, Ni. Wear-resistant coatings such as SiC; Ni. VIEE, Ni. C (graphite) anti-friction coating, etc.; weldable coatings such as Pb, Cu, Sn, Ag, etc.; anti-carburizing Cu coating, etc.
Material requirements
Coatings are mostly a single metal or alloy, such as titanium palladium, zinc, cadmium, gold or brass, bronze, etc.; there are also diffused layers, such as nickel-silicon carbide, nickel-fluorinated graphite, etc.; copper-nickel-chromium layer on steel, silver-indium layer on steel, etc. In addition to iron-based cast iron, steel and stainless steel, electroplating base materials also include non-ferrous metals, or ABS plastics, polypropylene, polysulfone and phenolic plastics, but plastics must undergo special activation and sensitization treatments before electroplating.
A technology that uses the principle of an electrolytic cell to deposit a metal coating with good adhesion but different properties and substrate materials on mechanical products. The electroplating layer is uniform than the hot-dip layer, and is generally thinner, ranging from a few microns to tens of microns. Through electroplating, decorative protection and various functional surface layers can be obtained on mechanical products, and workpieces that are worn and processed incorrectly can also be repaired.
In addition, common electroplating includes: copper plating, nickel plating, silver plating, gold plating, chrome plating, galvanizing, tin plating, vacuum plating, etc.
Different metal surface plating requirements also have different effects. Examples are as follows:
a. Copper plating: for primer, to improve the adhesion and corrosion resistance of the electroplating layer. (Copper is easy to oxidize. After oxidation, the verdigris no longer conducts electricity, so copper-plated products must be protected by copper)
b. Nickel plating: used as a primer or as an appearance to improve corrosion resistance and wear resistance (among them, chemical nickel is more wear-resistant than chrome plating in modern technology). (Note that many electronic products, such as DIN heads and N heads, no longer use nickel as the backing, mainly because nickel is magnetic, which will affect the passive intermodulation in the electrical performance)
c. Gold plating: improve conductive contact resistance and enhance signal transmission. (Gold is the most stable and the most expensive.)
d. Palladium nickel plating: improve conductive contact resistance, enhance signal transmission, and have higher wear resistance than gold.
e. Tin-lead plating: improve soldering ability, and will soon be replaced by other substitutes (because of lead content, most of them are changed to bright tin and matte tin).
f. Silver plating: improve conductive contact resistance and enhance signal transmission. (Silver has the best performance, is easy to oxidize, and conducts electricity after oxidation)
Electroplating is a method of covering a conductor with a layer of metal using the principle of electrolysis.In addition to electrical conductors, electroplating can also be used on specially treated plastics.
Process flow of electroplating solution formula for aluminum parts:
High-temperature weak alkali etching→cleaning→pickling→cleaning→zinc dipping→cleaning→secondary zinc immersion→cleaning→pre-copper plating→cleaning→pre-silver plating→cyanide bright silver plating→recycling wash→cleaning→silver protection→cleaning → Dry.
From the perspective of the process flow, the selected protective material must be resistant to high temperature (about 80°C), alkali-resistant, and acid-resistant. Secondly, the protective material can be easily peeled off after silver plating.
The protective materials sold in the market include peelable rubber, peelable paint, general adhesive tape and adhesive tape. The properties of acid resistance, alkali corrosion resistance, high temperature resistance (the maximum temperature of alkali etching solution is about 80°C) and peelability of these protective materials were tested respectively.

Electrophoresis
Electrophoresis (electrophoresis, EP) is the abbreviation of electrophoresis phenomenon, which refers to the phenomenon that charged particles move toward the electrode opposite to its electric property under the action of an electric field. The technique of using charged particles to move at different speeds in an electric field to achieve separation is called electrophoresis.Electrophoresis has been increasingly widely used in various fields such as analytical chemistry, biochemistry, clinical chemistry, toxicology, pharmacology, immunology, microbiology, food chemistry, etc.
According to different separation principles, electrophoresis can be divided into zone electrophoresis, boundary shift electrophoresis, isotachophoresis and focusing electrophoresis. According to whether electrophoresis is carried out in solution or on a solid support, it is divided into free electrophoresis and support electrophoresis.The electrophoresis methods used can be roughly divided into three categories: microelectrophoresis, free interface electrophoresis and zone electrophoresis. Zone electrophoresis is widely used.
Principle of electrophoresis:
Electrophoresis is the electrophoretic coating applied to the positive and negative poles. Under the action of voltage, the charged coating ions move to the cathode, and interact with the alkaline substances generated on the surface of the cathode to form insoluble matter, which is deposited on the surface of the workpiece. It includes four processes:
Electrolysis
(Decomposition) At the beginning of the cathode reaction, it is an electrolysis reaction, which generates hydrogen and hydroxide ions OH-. This reaction causes a highly alkaline boundary layer to be formed on the cathode surface. When the cation and hydroxide react to become insoluble in water, the coating film Deposition, the equation is: H2O→OH-+H+.
Electrophoretic movement
Swimming and migration Cationic resin and H+ move to the cathode under the action of an electric field, while anions move to the anode.
Electrodeposition
(Precipitation) On the surface of the coated workpiece, the cationic resin reacts alkalinely with the surface of the cathode, neutralizes and precipitates insoluble matter, which is deposited on the coated workpiece.
Electroosmosis
(Dehydration) The coating film on the surface of the coating solid and the workpiece is translucent, with a large number of capillary pores, and water is drained from the cathode coating film. Under the action of the electric field, the coating film is dehydrated, and the coating film is adsorbed. on the surface of the workpiece to complete the entire electrophoresis process.

Passivation
Passivation, also known as chromate treatment, is a pickling process that removes surface grease, rust, and oxides by immersion or ultrasonic cleaning. Through the chemical reaction of the passivation solution, it can prevent corrosion and prolong rust. The color of the passivation film will change with different materials. Passivation will not increase the thickness of the product, and there is no need to worry about it affecting the accuracy of the product.
After the metal is treated with an oxidizing medium, its corrosion rate is significantly lower than that before the original untreated phenomenon, which is called the passivation of the metal. The passivation mechanism can be mainly explained by the thin film theory, that is, the passivation is due to the interaction between the metal and the oxidizing medium, and a very thin, dense, good covering performance, which can firmly adhere to the metal surface, is formed on the metal surface. Passive film on the surface. This film exists as a separate phase, usually a compound of oxygen and metal. It plays the role of completely separating the metal from the corrosive medium, preventing direct contact between the metal and the corrosive medium, so that the metal basically stops dissolving and forms a passive state to prevent corrosion.
Passivation treatment operation method: the process of using chromate solution and metal to form a trivalent or hexavalent chromium layer on the surface is called passivation, also known as chromization. It is mostly used in the treatment of aluminum, magnesium and their alloys. It can also form a chromium layer on steel, but it is rarely used alone. It is often used in conjunction with phosphating to close the pores of the phosphating layer and passivate the exposed steel in the phosphating layer. Phosphate, to inhibit the corrosion of residual phosphating accelerator, and further increase the protection ability. Potassium dichromate solution (2-4 g/L, sometimes 1-2 g phosphoric acid is added) is generally used for passivation, soaked at 80-90 degrees Celsius for 2-3 minutes, taken out, and washed with water.In the process of etching stainless steel, we often encounter yellowing of the product, here we need passivation process to deal with it.

Blackened
Blackening is also called bluing. The principle is to immerse the product in a strong oxidizing chemical solution to form an oxide film on the metal surface to isolate the air and achieve the purpose of rust prevention. This process is applicable to steel materials.
The commonly used methods of blackening treatment include traditional alkaline heating blackening and room temperature blackening that appears later. However, the normal temperature blackening process is not very effective for low carbon steel. Alkaline blackening is subdivided, and there is a difference between one blackening and two blackening. The main components of the blackening solution are sodium hydroxide and sodium nitrite. The temperature difference required for blackening is not large, and a good surface can be obtained between 135-155°C, but the time required is somewhat long.
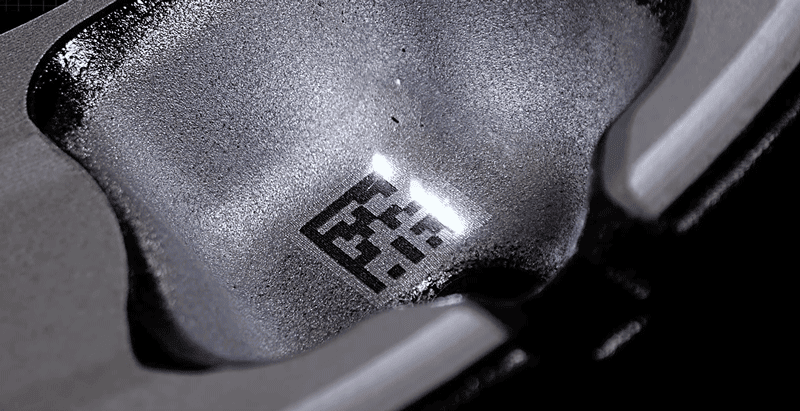
Laser engraving
Laser engraving is also called laser engraving or laser marking. Laser engraving is based on numerical control technology and laser is the processing medium. The physical denaturation of the instant melting and gasification of the processed material under laser irradiation achieves the purpose of processing.
Laser processing features: no contact with the surface of the material, not affected by mechanical movement, the surface will not be deformed, generally no need to fix. Not affected by the elasticity and flexibility of the material, it is convenient to process soft materials. High processing precision, fast speed, wide range of applications.The laser engraving effect is permanent, the surface quality is high, and it is suitable for products made of various metal and plastic materials.

Silk screen
Silk screen printing means that the ink transfers the pattern to the product through the screen. The color of the ink can be customized according to the needs of customers. DD Prototype has done 6 colors on the same product, including black, red, blue, yellow, White green. If you want the effect of silk screen printing to be more durable, you can also add a layer of UV after silk screen printing to prolong its life. Silk screen printing is suitable for various metal and plastic materials, and can also be combined with surface treatment such as oxidation, painting, powder spraying, electroplating, and electrophoresis.

Polishing
Polishing is to make the product beautiful, translucent and protect the surface. Polishing and transparency is a good choice for you. The polishing of hardware products is divided into manual polishing, mechanical polishing, and electrolytic polishing. Electrolytic polishing can be used to replace heavy mechanical polishing, especially for parts with complex shapes and parts that are difficult to process by manual polishing and mechanical methods. Electrolytic polishing is often used for steel, aluminum, copper and other parts.

Brushing
Brushing is a surface treatment method that forms lines on the surface of the workpiece through a flat-pressed abrasive belt and a non-woven roller brush to achieve a decorative effect. Brushed surface treatment can reflect the texture of metal materials, and it is becoming more and more popular in modern life. It is widely used in mobile phones, computers, monitors, furniture, electrical appliances and other shells.
Power Coating, Painting
Power Coating and Painting are two common surface treatments in hardware parts spraying, and they are the most commonly used surface treatments for precision parts and small batch customization. They can protect the surface from corrosion, rust, and can also achieve an aesthetic effect. Both Power Coating and Painting can be customized with different textures (fine lines, rough lines, leather lines, etc.), different colors, and different gloss levels (matte, flat, high-gloss).
Teflon Coating
Also known as Teflon spraying, it is a very individual surface treatment. It has excellent anti-adhesiveness, non-stickiness, high temperature resistance, low friction, high hardness, non-humidity, and high chemical resistance. It is widely used in the food industry. , tableware, kitchen utensils, paper industry, medical equipment, electronic products and automotive products, chemical equipment, etc., while protecting the material from chemical corrosion and prolonging the service life of the product.
Sand blasting
Sand blasting is a process for surface treatment of workpieces. Compressed air is used as the power to form a high-speed jet beam to spray the spray material (copper ore, quartz sand, corundum, iron sand, sea sand) onto the surface of the workpiece to be treated at high speed, so that the appearance or shape of the workpiece surface changes. Due to the impact and cutting effect of the abrasive on the surface of the workpiece, the surface of the workpiece can obtain a certain degree of cleanliness and different roughness, so that the mechanical properties of the surface of the workpiece can be improved, thus improving the fatigue resistance of the workpiece, increasing it and the coating The adhesion between them prolongs the durability of the coating film, and is also conducive to the leveling and decoration of the coating.